Turkish health regulators approved the Chinese Sinovac Covid-19 vaccine for emergency use on Wednesday, clearing the way for a mass vaccination program to begin on Thursday.
“The scientific data has been evaluated and 14-day testing of samples taken from the batches that have arrived have been examined in our laboratories... Emergency use authorization has been issued," the Turkish Drug and Medical Device Administration said in a statement.
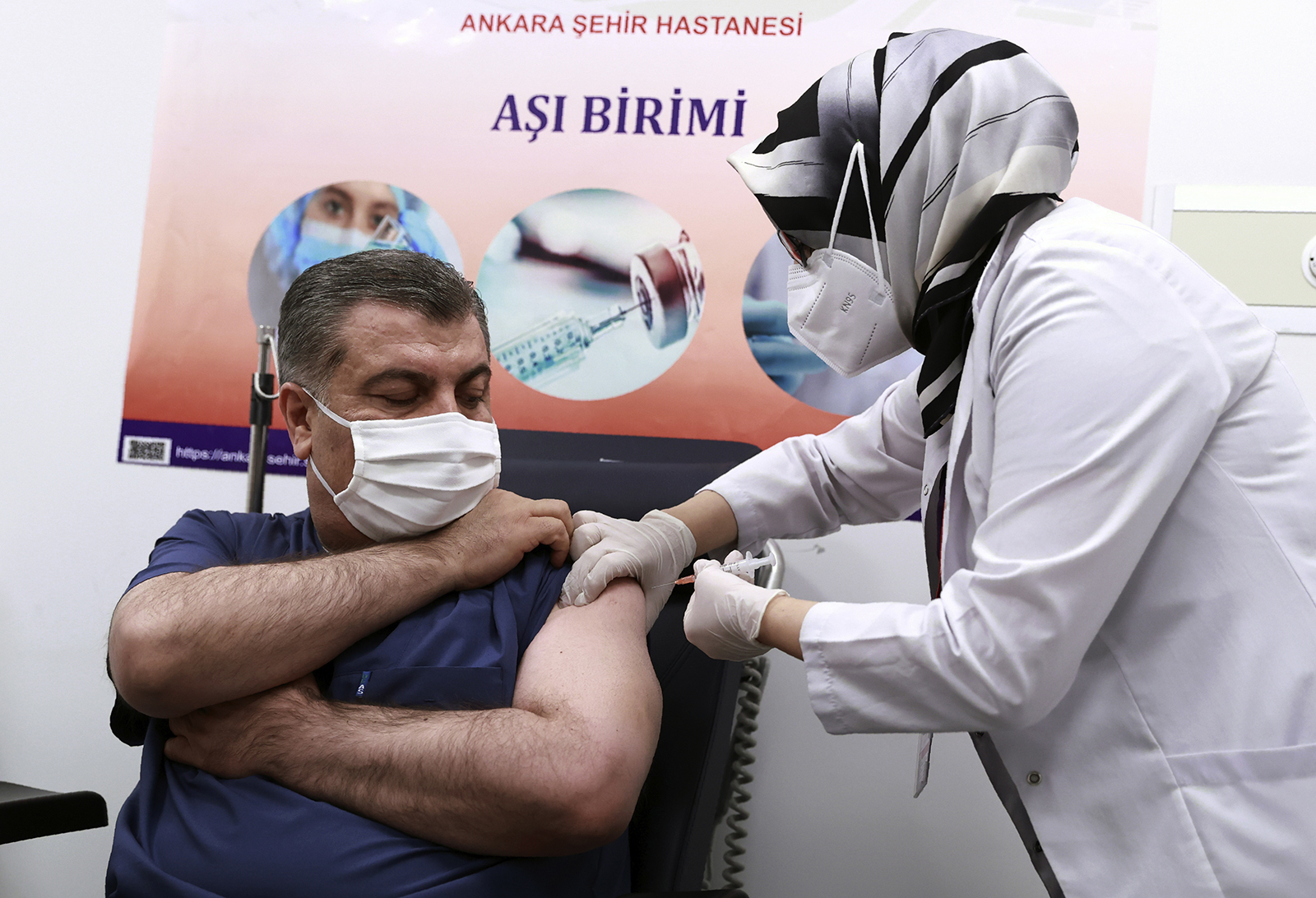
Immediately after the announcement, Turkish Health Minister Fahrettin Koca received the first dose of the vaccine on live television. He told reporters that he and other members of the Pandemic Science Council are being vaccinated immediately and rollout across all 81 provinces in Turkey will begin on Thursday.
The administration did not release details about the data used to determine the vaccine’s safety and efficacy. Advanced, Phase 3 trials are ongoing in Turkey, but the vaccine is also being tested in other countries, including Brazil.
Chilean President Sebastian Pinera announced on Wednesday that his country expects to receive 10 million doses of the Sinovac vaccine. Indonesian President Joko Widodo received the first shot of Sinovac vaccine on Wednesday and announced that vaccination will start across the country immediately.
Questions over Sinovac data: On Tuesday, the government of Sao Paulo published data showing that the Sinovac vaccine was just 50.38% effective in late-stage trials in Brazil, significantly lower than earlier results showed.
While the number exceeds the threshold required for regulatory approval, it falls far below the 78% previously announced.